Accelerated Evolution
We are a new lab at the Generative Biology Institute, EIT, Oxford, UK.
We aim to leverage an orthogonal replication system for the accelerated evolution of complex biological functions to:
advance fundamental scientific understanding of molecular and cellular evolution.
develop biomolecules to tackle challenges in human-health and industry, in collaboration with colleagues at GBI such as the Rehm group.
generate large-scale enzyme datasets for training next-gen protein and cell design models, in collaboration with colleagues at GBI such as the Jamali group.
Key Tech - EcORep
The evolution of new function in living organisms is the result of continuous genomic mutation and selection within a population. This process is slow, and the rate of evolution is fundamentally limited by the critical mutation rate. Directed evolution commonly sidesteps the limitation on in vivo mutation rate by generating genetic diversity in vitro, but this does not enable the continuous evolution of genes within an organism. The mutation rate of cells can be transiently increased, but high levels of untargeted mutation lead to a catastrophic mutational load on the genome and are unsustainable.
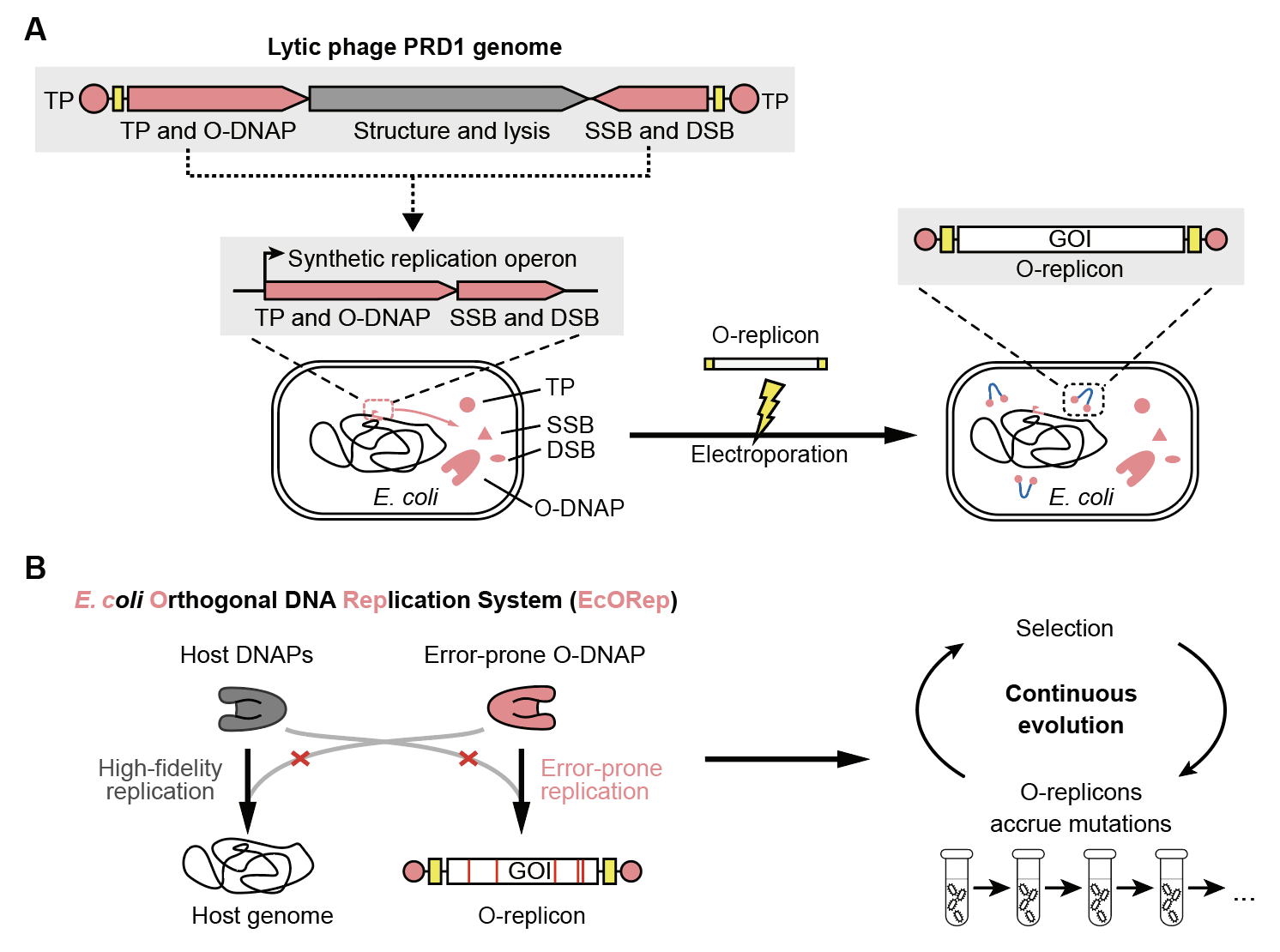
We established EcORep, an orthogonal replication system in Escherichia coli, the workhorse of molecular biology, using selected genetic elements from a lytic phage which replicates uncontrollably and lyses cells in less than 60 minutes (Figure 1A). The orthogonal replicon is not copied by host DNA polymerases, but is selectively copied by an orthogonal DNA polymerase (O-DNAP), which does not copy the genome. The orthogonal replicons can carry diverse DNA cargos and are not copied by host polymerases but are selectively copied by orthogonal DNA polymerases (O-DNAPs), which do not copy the genome (Figure 1B). We engineered mutant O-DNAPs that selectively increase the mutation rate of the orthogonal replicons and demonstrated the utility of the system for accelerated continuous evolution by rapidly evolving cellular phenotypes in multiple replicates. This technology enables the rapid development of diverse research tools, biopharmaceutical leads, and strains for the production of industrial chemicals.
References
Tian, R. et al. Establishing a synthetic orthogonal replication system enables accelerated evolution in E. coli. Science 383, 421–426 (2024).
Rix, G. et al. Continuous evolution of user‑defined genes at 1 million times the genomic mutation rate. Science 386, eadm9073 (2024).
Tian, R. et al. Engineered bacterial orthogonal DNA replication system for continuous evolution. Nat. Chem. Biol. 19, 801–810 (2023).
Molina, R. S. et al. In vivo hypermutation and continuous evolution. Nat. Rev. Methods Prim. 2, 36 (2022).